Welcome to the Early Psychosis Intervention Ontario Network (EPION) Metabolic Monitoring interactive tool!
This web tool aims to promote metabolic monitoring for people with mental illness. It provides a structured way to screen and begin metabolic interventions and defines the roles of members of the mental health and primary care teams.
Who can use this interactive web tool?
Community- and hospital-based psychiatrists, nurse practitioners, family physicians, and frontline mental health specialists working with people living with psychosis and families. Clients are also encouraged to use this web tool to track their own metabolic risk factors and set related healthy lifestyle goals
Who designed this tool?
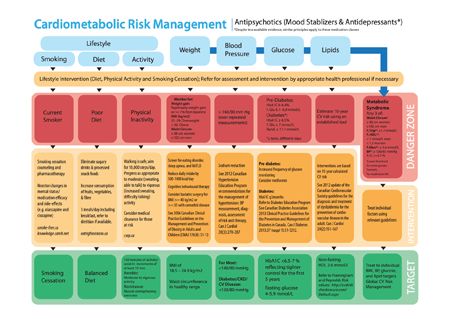
The EPION Metabolic Monitoring Working Group developed this web tool based on its own metabolic monitoring resources, including the Cardiometabolic Risk Management tool,* which was revised according to Canadian guidelines by EPION and its partners. The partners include the Centre for Addiction and Mental Health, Ontario Shores Centre for Mental Health Sciences, The Royal Ottawa Mental Health Centre, and Waypoint Centre for Mental Health Care, with thanks to the On Track First Episode Psychosis Program for their collaboration.
*Adapted for use by the EPION Metabolic Monitoring Working Group, with permission from Curtis, J., Newall, H., Samaras, K. © HETI 2011; Shiers, D et al.
The original versions of the resources used to develop this interactive web tool are available for download here.
How to use this tool?
The interactive web tool does not collect any personal health information. Once you have entered all the necessary information, you can print a copy or save it to your computer.
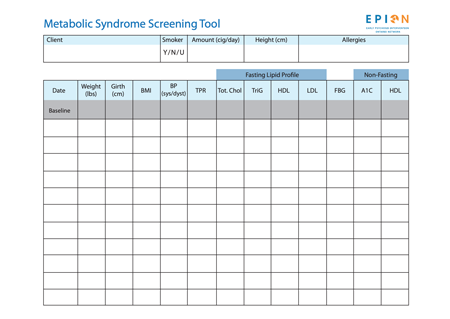
This interactive web tool allows clients and clinicians to monitor the following:
-
measurements (waist, blood pressure, HDL cholesterol, triglycerides (TG), blood sugar, and body mass index (BMI).
-
medications and current side effects
-
lifestyle (smoking, alcohol use, exercise)
-
sleep
-
nutrition.
As you enter the information, a “danger zone” pop-up will appear if there are any areas of concern. This pop-up will include suggestions for healthy target zones, and you can also include healthy lifestyle goals that you want to achieve.
The healthy lifestyle goals will also appear at the bottom of the web tool so that you can create additional goals. You will also be able to add reminders to your calendar so you can visit the tool again in the future to continue monitoring and following up on your goals.
How do I use EPION’s metabolic monitoring resources?
Three short instructional videos on how clinicians can use the original versions of resources used to develop this tool, are available here:
-
An overview of the suite of tools:
-
Using the EPION Metabolic Syndrome Screening Tool:
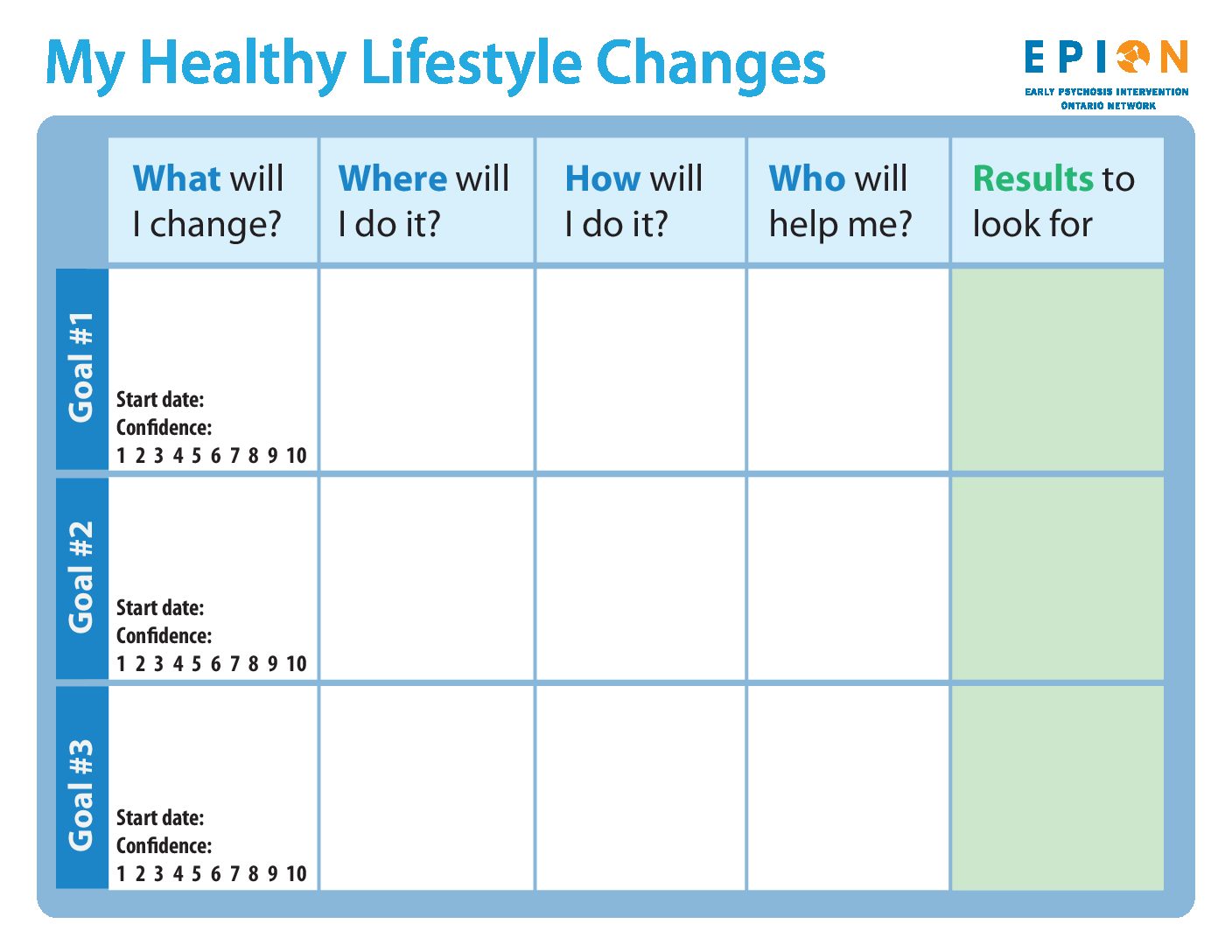
Goals
The interactive web tool allows you to set healthy lifestyle goals or any other goals you’d like to focus on. These don’t have to be big goals – small steps can also be effective! Just enter your goal, as well as where, how and what you need to do to achieve it. List all the people who can help you achieve this goal, and the milestones you can use to find out if you’re reaching your goal.
You can also include the start date and your confidence level, and add personal calendar reminders so you can come back to the web tool to monitor your progress.
My Healthy Lifestyle Changes
Follow-up
This section allows you to create a follow-up list, including questions you may have for your care team about metabolic monitoring, or items in this assessment that they can help your complete. You can print out this list and bring it to your next appointment with your care team.
Monitoring: How Often and What to Do
Applies to patients prescribed antipsychotics and metabolically active mood stabilizers and antidepressants.
Frequency: As a minimum review those prescribed a new agent at baseline and at least once after 3 months. Weight should be assessed monthly in the first 3 months of taking a new antipsychotic as rapid early weight gain may predict severe weight gain in the longer term. Subsequent review should take place annually unless an abnormality of physical health emerges, which should then prompt appropriate action and/or continuing review at least every 3 months.
History: Ask about family history (diabetes, obesity, CVD in first degree relatives <60 yrs), gestational diabetes. Note ethnicity.
| Baseline | 4 weeks | 8 weeks | 12 weeks | Quarterly | Annually | |
|---|---|---|---|---|---|---|
| Personal/FHx | X | X | ||||
| Lifestyle Review1 | X | X | X | X | X | X |
| Weight/WC/ | X | X | X | X | X | X |
| BP | X | X | X | |||
| FPG/HbA1C | X | X | X | |||
| Lipid Profile 2 | X | X | X |
1 Smoking, diet, and physical activity. 2 If fasting lipid profile cannot be obtained, a non-fasting sample is satisfactory
Derived from consensus guidelines 2004, j clin. psych 65:2
Information for GPs
Investigations: Fasting estimates of plasma glucose (FPG), HbA1c, and lipids (total cholesterol, non-HDL, HDL, triglycerides). If fasting samples are impractical, then non-fasting samples are satisfactory for most measurements except for LDL and triglycerides.
Review of Antipsychotic Medication
Choose lower metabolic liability medication first-line when possible. Response in first episode psychosis is robust independent of agent.
Changing or discontinuing antipsychotic requires careful clinical judgment, balancing metabolic benefits against relapse risk. Ideally psychiatrist supervised. Should be a priority if there is:
-
Rapid weight gain (e.g. 5kg <3 months) following antipsychotic initiation.
-
Rapid development (<3 months) of abnormal lipids, BP, or glucose.
The psychiatrist should consider whether the antipsychotic drug regimen has played a causative role in these abnormalities and, if so, whether an alternative regimen could be expected to offer less adverse effect:
If clinical judgment and patient preference support continuing with the same treatment then ensure appropriate further monitoring and clinical considerations. Avoid antipsychotic polypharmacy when possible; Avoid off-label use of antipsychotics.
Specific Adjunctive Pharmalogical Interventions
For mitigation of excess weight gain associated with antipsychotic use, the strongest evidence is for off-label use of Metformin and Topiramate (Mayan, 2010). Consider Metformin first due to better tolerability profile unless there is a co-morbid bingeeating disorder (McElroy, SI, 2009). Please be advised that off-label use requires documented informed consent. Discontinue if no sign of efficacy (continuing weight gain if used for weight loss or stabilization) after 3 months at therapeutic dose.
Metformin:
Weight Gain & Primary Prevention of Diabetes: Start 250 mg po BID, titrate every 1-2 weeks as clinically indicated and tolerated. Dose range is 750 mg – 2 Gm/day
Caution with renal or hepatic impairment. Avoid excessive alcohol use. Monitor for GI side effects. Monitor for B12 deficiency.
Topiramate:
Weight Gain: Start 12.5 mg po BID; titrate by 25-50 mg per day in divided doses every 1-2 weeks as tolerated to a maximum of 100 mg po BID.
Caution with renal or hepatic impairment. Avoid excessive alcohol use. Monitor for cognitive changes. Parasthesias are common but generally well-tolerated.